Lukman Ismaila, Fellow, Datasphere Initiative
Introduction
Medical data plays a crucial role in scientific research and in the ability of the outputs of this research to impact real-world medical care, as it allows researchers to better understand the relationships between various factors such as genetics, environmental exposures, and health outcomes, and later allow medical care practitioners to have better tools for diagnosis and care.
In addition to its use in clinical trials, medical data is also important for a wide range of other research efforts. For example, data can be used to identify patterns of disease prevalence and incidence in different populations, to investigate the relationship between genetics and health outcomes, and to understand the environmental and social factors that may influence health.
Overall, the importance of medical data in scientific research cannot be overstated. It is a crucial resource that allows researchers to make informed decisions and to develop new treatments and interventions that can improve the health and well-being of individuals around the world.
One key area where medical data is particularly important is in clinical trials, where it is used to evaluate the effectiveness and safety of new treatments. By collecting data from a large number of patients, researchers can determine whether a new treatment is likely to be effective and whether it has any potential side effects. This information is essential in determining whether a new treatment should be approved for widespread use. By analyzing large datasets, researchers can identify patterns and trends that may not be immediately apparent when looking at individual cases. This later can help to inform the development of new treatments and interventions, as well as to improve our understanding of disease mechanisms and risk factors.¹
However, in the African continent, some enabler elements to inform this virtuous value chain are far from ideal, such as the availability and quality of data for medical research purposes.² Thus, throughout my Datasphere Initiative fellowship, I will identify and investigate the challenges that are holding Africa back in this regard, including issues such as gaps in technology (e.g., from internet connectivity to Health Information System – HIS), digital and data literacy, enabling policies and governance models that core actors in the health sector could follow. My lens into this problem is medical imagery as data.
If these challenges are not tackled, the hope of using data to drive sustainable development, especially in health, remains far from ideal if better data policies and efficiency are not in place.
Challenges of medical data unavailability in the context of scientific research in Africa
Although recent improvements in digital technology enable better ways to collect and manage data for research purposes, including in health, research still experiences significant limitations in access to data, especially in Africa.³ In respect to medical imagery data, access is not only the problem, but existence in first place of data also poses a challenge. An example is medical imagery for rare diseases. Other access challenges include limited clinical expertise for medical data preprocessing and stringent policy on data sharing by various government agencies and international actors.
This is not a new discussion across the continent and within the international community. Already in 2015, a workshop on Sharing Research Data to Improve Public Health in Africa concluded that more and better data should be generated in and to Africa. From that call, various international and African expert networks were formed to tackle the challenges identified then, including H3Africa, INDEPTH, and the ALPHA network. These efforts are focused on empowering and tackling the challenges faced by African countries.⁴
Advances can also be observed when one sees a constant increase of studies available that report on data sharing among health researchers in Africa, as shown in figure 1 and reported by Obiora et al. in their effort of mapping the existing evidence regarding the data-sharing practices of health researchers in African countries.⁵
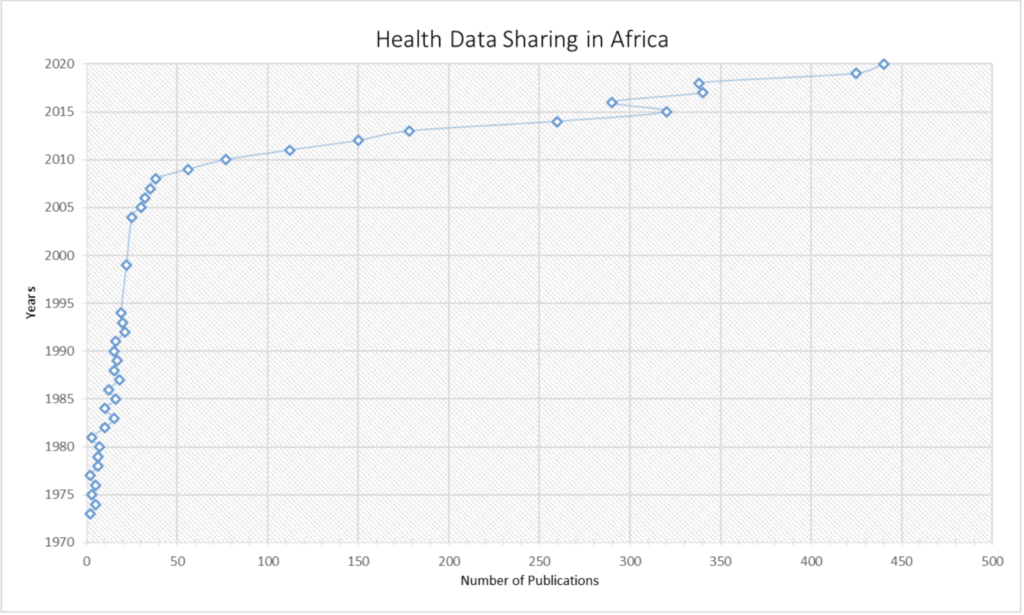
However, several known factors still contribute to the unavailability of good data for scientific research in Africa. Some of these include:
- Cross-border transfers of personal data and regulatory complexities: Cross-border data is a challenge in every sector, but especially in a highly regulated sector such as health. However, to solve some of the most pressing health challenges, global collaborations and thus cross-border data sharing might be required. The challenge is that in Africa one can still observe a lack of regulation, and regulatory fragmentation, a lack of meaningful data protection statutes, and the absence of independent regulators dedicated to the execution of the existent privacy laws.⁶
- Limited funding: Many research institutions in Africa have limited funding, which makes it difficult to invest in the infrastructure and resources needed to collect, store, protect and analyze large datasets. This significantly limits the ability of researchers to generate and use medical data in their work in a sustainable way.
- Poor data management: Many research institutions in Africa lack robust data management systems and practices, which eventually makes it difficult to collect, store, analyze, replicate and share data effectively. This also can hinder the use of medical data in research efforts.
- Limited access to technology: In some parts of Africa, access to technology may be limited, which makes it difficult for researchers to collect and analyze data using modern technology like MRI and CT scanners for high-resolution medical image acquisition. Challenges in connecting institutions at the technology level (e.g. lack of interoperability) also create barriers for access and sharing and lastly, unavailability of reliable technology for secure storage of medical images can lead to data loss and/or corruption.
- Cultural and ethical considerations: A number of cultural and ethical considerations in different regions make it difficult to collect certain types of medical data in Africa. Beyond the traditional lack of trust in some systems that collect and store data or in some cases lack of trust in government or international or local institutions, in Africa, some communities might be against data collection due to specific cultural or religious beliefs.
Overall, the lack of availability of medical data for scientific research in Africa is a complex issue that is influenced by a range of factors. Addressing these challenges will require a multi-faceted approach that includes investments in infrastructure, training, and technology, as well as efforts to address cultural and ethical considerations.
Conclusion and next research steps
As one can see, these challenges are not easy to tackle, and thus multifaceted approaches are needed. Some of these approaches might include the implementation and maintenance of well-articulated data policies and standards, the development of mechanisms for oversight and accountability, fostering of a culture of data sharing, and increased infrastructure and technology investment. I will further explore these suggestions during my fellowship, applying the concept of the Datasphere to my analysis.
The Datasphere Initiative is focused on the development of agile frameworks to safely unlock the potential of data for all by a worldwide network of stakeholders supporting a comprehensive and creative approach to data governance. By cataloging and categorizing challenges under the three pillars – data, norms, and people – we can devise solutions.
¹ Mamuye, Adane L., et al. “Health Information Exchange Policy and Standards for Digital Health Systems in Africa: A Systematic Review.” PLOS Digital Health, vol. 1, no. 10, Oct. 2022, p. e0000118. PLoS Journals, https://doi.org/10.1371/journal.pdig.0000118.
² Staunton, Ciara, et al. “Data Protection, Data Management, and Data Sharing: Stakeholder Perspectives on the Protection of Personal Health Information in South Africa.” PloS One, vol. 16, no. 12, 2021, p. e0260341. PubMed, https://doi.org/10.1371/journal.pone.0260341.
³ “Health Data Privacy and Data Sharing Are Possible in Africa.” The Guardian Nigeria News – Nigeria and World News, 26 Oct. 2022, https://guardian.ng/apo-press-releases/health-data-privacy-and-data-sharing-are-possible-in-africa/
⁴ Committee on Population, et al. Sharing Research Data to Improve Public Health in Africa: A Workshop Summary. National Academies Press (US), 2015. PubMed, http://www.ncbi.nlm.nih.gov/books/NBK321547/.
⁵ Obiora, Oluchukwu Loveth, et al. “Data Sharing Practices of Health Researchers in Africa: A Scoping Review Protocol.” JBI Evidence Synthesis, vol. 20, no. 2, Feb. 2022, p. 681. journals.lww.com, https://doi.org/10.11124/JBIES-20-00502.
⁶ Examples of existent privacy laws that impact medical personal data include, for instance, the Protection of Personal Information Act (POPIA) in South Africa ² or the National Data Protection Regulation (NDPR in Nigeria). In Nigeria, a regulatory framework for data protection is anticipated to be reinforced with the passage of the draft Data Protection Bill 2022 by the Data Protection Bureau.⁷
⁷ Babalola, Olumide. “The National Data Protection Authority in Nigeria: A Critical Examination of the Nature and Enforcement Mechanism of National Information Technology Development Agency (NITDA) under the Nigeria Data Protection Regulation 2019 and Its Enabling Act.” SSRN Electronic Journal, 2021. DOI.org, https://doi.org/10.2139/ssrn.4315937.




